Can Your Gut Bacteria Impact Your Weight? (And What To Do About It)
How gut bacteria extract calories, control appetite hormones, and determine whether your body stores or burns fat; A practical guide to create the gut environment that supports your healthiest weight
You’ve probably heard that gut health matters for weight loss. What you might not realise is that the 100 trillion bacteria living in your intestines play an active role in deciding whether you burn fat or store it or whether you feel satisfied after a meal or keep opening the fridge.
This isn’t theoretical. We have actual scientific proof that our gut microbes influence far more than we ever thought.
Take this for example: in a 2025 study, adolescents with obesity who received faecal microbiota transplants taken from lean donors (that’s an ingested poop sample, from one individual to another, yes) showed significant differences four years later compared to those who received a placebo - 10cm smaller waist circumference, nearly 5% less body fat, and 68% lower inflammation markers. The transplanted bacteria created lasting metabolic changes. In another recent trial, over half the participants who received a transplant of bacteria into their gut, achieved significant weight loss averaging nearly 8kg within 12 weeks.
The bacteria in our gut don’t just influence weight - they actively control how our bodies process and store energy.
Our gut bacteria extract calories and store fat
The bacteria in our gut determine how much energy we harvest from food. One study showed that obese individuals have a different bacterial composition than lean people - specifically, a higher ratio of one kind of bacteria compared to others, which allows them to extract up to an additional 150 calories per day from the same food. Another study found that people whose microbiomes bacteria generated more methane gas tend to extract more energy from high-fiber foods.
The mechanisms are not yet completely clear, but a consensus is being built that the same meal can yield different calorie amounts depending on your gut bacteria. With the additional calories per day example, extrapulated over a month, we are looking at 4,500 extra calories. Over a year, it’s enough to explain significant weight gain without changing the diet at all. Facinating!
And there is more. Your body is constantly making decisions about whether to store incoming energy as fat or burn it. Certain bacteria suppress a protein that normally tells your body to burn fat rather than store it (fasting-induced adipose factor or FIAF). When gut bacteria shut down that signal, your fat cells default to storage mode - even if you’re not overeating.
If gut bacteria are out of balance, meaning not enough ‘beneficial’ bacteria and too much ‘less beneficial’ bacteria, the protective barrier lining the gut can weaken. As that barrier weakens, inflammatory compounds leak into the bloodstream and can trigger insulin resistance - which makes the body store fat more easily and burn it less readily. This can also creates constant hunger, because cells aren’t getting energy efficiently, even when blood sugar is high.
So while the basic concept of calories in and calories out will always hold, there is more to the story. Our gut composition can make it harder or easier to lose weight and it can impact how hungry we are (and hence how many calories we will find ourselves driven to eat), as well as how many calories we absorb from the same meal.
Overwhelmed much?
Ok, at this point it would be fair to feel like you have no control over any of it. I know I did at times. You might be thinking, ‘I am being asked not only to eat a certain amount of calories, aim for healthy sources, and I can’t even control how that plays out? Off to the cookie jar I go! ‘
But here is how I think about it:
The basics always hold. You’ll lose weight if you ingest fewer calories than you burn. That formula will always be true. But that formula has many things impacting it - how much you absorb depends on your gut bacteria, how hungry or satisfied you are affects how much you eat, and what you eat impacts how you store or burn fat. The environment you create through what you eat, when and in what quantities, how you sleep and how much you move will either make the weight loss process smooth and easy or an uphill struggle.
And most importantly, you’re aiming for a healthy weight to feel happy and healthy, to thrive. The weight is a byproduct of helping your body perform at its best. To help your body thrive, all these moving factors that support health are important as much (if not more) than the weight itself.
Deep breath and let’s get back to our gut bacteria and how fuelling them can help.
Short-chain fatty acids control your appetite and metabolism
Beneficial bacteria in our gut ferment the fibre we eat and produce short-chain fatty acids (acetate, propionate, and butyrate). These molecules trigger the release of GLP-1 and PYY - two powerful appetite-suppressing hormones. GLP-1 is the same hormone that weight-loss medications like Ozempic mimic.
GLP-1, which is naturally excreted in our body post meals, slows how quickly food leaves the stomach, making us feel full longer.
It also tells our brain when we have had enough to eat. Butyrate, one of those short-chain fatty acids bacteria produces, directly increases the production of PYY, a hormone that signals fullness. The more butyrate your bacteria produces, the stronger that satiety signal becomes.
So the more fibre we consume, the ‘happier’ our gut bacteria, the more short-chain fatty acids we produce and the higher satiety we feel. This is one of the reasons high-fibre diets are so important for feelings of satiety and reduced hunger.
But here’s where it gets interesting for weight loss: not all fibre works the same way, and not all gut bacteria do the same job.
This is why diversity of fibre sources matters. A healthy microbiome isn’t just about having beneficial bacteria - it’s about having enough different types working together. Some bacteria specialise in producing short-chain fatty acids that signal fullness. Others maintain the gut lining that allows those compounds to be absorbed effectively. And others help regulate how your body responds to hunger and satiety signals. When this ecosystem is disrupted, you might be eating plenty of fiber but not getting the full metabolic and appetite-regulating benefits.
The key takeaway: it’s not just enough to feed our existing bacteria by consuming fibre - we need to cultivate a diverse population of bacteria by eating diverse fibre sources. That’s how we support the full range of mechanisms that help with sustainable, comfortable weight loss.
For introduction of new bacteria strains, fermented foods are as important as fibre
The recommendation is clear: eat more fibre to feed your gut bacteria. While this recommendation holds, a 2021 study from Stanford added some important nuance.
They split participants into two groups. One increased fibre from 22 to 45 grams daily. The other ate six servings of fermented foods daily - yogurt, kefir, kimchi, sauerkraut, kombucha, vegetable brine drinks.
After 10 weeks, the high-fibre group showed no increase in bacterial diversity. The fermented foods group showed significant increases in both diversity and number of bacterial species. Even more interesting, 19 inflammatory proteins (bad for us) decreased in the fermented foods group, including IL-6, which is linked to type 2 diabetes and chronic inflammation.
The researchers found undigested carbohydrates in the stool of the fibre group, suggesting their depleted microbiomes (not diverse enough) didn’t have the needed bacteria to ferment the fibre.
The practical implication: you may need to repopulate the bacteria first through fermented foods that introduce new bacterial strains, and only then can those bacteria utilise the fibre you’re eating.
How to increase diversity? Eat a variety of plant foods
The American Gut Project analysed over 10,000 participants and found that people eating 30 or more different plant types weekly had significantly more diverse gut microbiomes than those eating fewer than 10. This held true whether they were vegan, vegetarian, or omnivore.
Here is what they counted toward 30: vegetables, fruits, whole grains, legumes, nuts, seeds, and herbs or spices. Different varieties count separately - red apples and green apples are two different plants.
High-diversity eaters showed elevated levels of Faecalibacterium prausnitzii, a major anti-inflammatory bacteria, and Oscillospira, a bacteria which is associated with leanness.
The mechanism behind this comes down to polyphenols - the plant compounds that give foods their colours. About 90-95% of polyphenols reach our colon unabsorbed, where they act as fertilisers for beneficial bacteria as well as inhibiting harmful ones.
Polyphenol-rich foods are usually plant-based foods with strong colours. They include berries (blueberries, cranberries, blackberries, strawberries), grapes, green tea, dark chocolate (70% cocoa or higher), extra virgin olive oil, and coffee. Herbs and spices are particularly concentrated sources - oregano, thyme, rosemary, turmeric, and cinnamon. Nuts like walnuts and pecans, along with colourful vegetables like red cabbage, artichokes, and spinach, also contribute significant amounts as well as apples, citrus fruits, and red onions.
This is part of why you will often hear the recommendation to ‘eat the rainbow’ and make sure you eat a variety of colourful vegetables and fruits. If you include several of these colourful foods throughout the day: a cup of berries on your yogurt, green tea with lunch, extra virgin olive oil on your salad, and dark chocolate as an occasional treat - you are sorted.
Some bacteria are consistently linked to healthy weight
Akkermansia muciniphila, a certain type of bacteria, has recently become popular as a promising weight-management bacteria, making up 1-5% of healthy gut bacteria but significantly depleted in obesity. It has been shown to strengthen the gut barrier, stimulate GLP-1 production, and reduce the inflammatory compounds that drive insulin resistance.
A 2024 trial showed that taking it as a supplement reduced body weight, fat mass, and blood sugar levels - but only in participants who had low levels to begin with. If you already had adequate Akkermansia levels, adding more doesn’t seem to help. Which makes it tricky, as most of us don’t know the exact composition of our gut bacteria and don’t test for levels unless something is off.
What we can do instead is rely on what we know boosts Akkermansia levels. In mice trials as well as some human ones, cranberry extract dramatically increased Akkermansia and prevented diet-induced obesity.
Another interesting bacteria is Christensenella minuta, which is dubbed the “lean bacteria” because it’s consistently more abundant in normal-weight individuals. Cornell researchers transplanted it into germ-free mice and reversed their weight gain. It produces high levels of acetate, inhibits fat production in the liver, and functions as a keystone species that helps other beneficial bacteria thrive.
Who knows, maybe at some point in the future we might have clear menu options and preferences on what to eat to boost specific types of bacteria. For now, our best bet is to ‘eat the rainbow’: a variety of colourful fruits and vegetables, rich in fibre and consume fermented foods, consistently.
What doesn’t work so well: some artificial sweeteners, highly processed foods and restriction
There is mixed data on artificial sweeteners. Saccharin and sucralose have been shown to alter the microbiome in ways that trigger glucose intolerance, making blood sugar control worse and reducing diversity. Aspartame’s effects are less clear, but some research found it to be “not benign for the microbiome,” reducing bacterial richness.
The bottom line is that we don’t yet know the exact impact of artificial sweeteners on the gut, but moderation in consumption is likely a good recommendation. Like everything in nutrition - it depends. If you are currently consuming full sugar drinks like Coke, regularly, the negative impact on your metabolism, weight and health probably outweighs the negative impact of a diet coke with aspartame on your gut…
Non-synthetic options like stevia, monk fruit and xylitol seem to be less disruptive, but the data is still emerging.
Restrictive dieting
Restrictive dieting (not eating enough calories or restricting specific nutrients) create a different problem. The dieting may create lasting microbiome changes, due to the lack of enough nutrient sources needed for bacteria to thrive.
Research in mice suggests the microbiome may retain changes after weight loss, with one study showing an altered bacterial composition for approximately six months post-diet. The mice rapidly regained weight when returning to normal eating, not because they started to eat more, but due to bacterial changes in the gut.
If true for humans too (we don’t know), it suggests that the quality of our diet during and after weight loss matters as much as the weight loss itself. Each round of restrictive dieting could deplete beneficial bacteria that take months to repopulate. The microbiome damage could potentially accumulate faster than it can repair between diets, which is why we should always be eating for health first, with weight loss as a secondary outcome.
So what do I eat?
Can we counter each and every new development about what happens to our gut microbiome when it relates to weight loss? Probably not. But what’s clear is that the composition of the gut microbiome has an impact on our ability to maintain a healthy weight and that good diversity of bacteria and a flourishing gut microbiome is beneficial in so many ways.
What might help?
Include fermented foods regularly - Consider adding some source of fermented food to your day - ideally from different sources. This could be yogurt or kefir, kimchi or sauerkraut. Mixing your sources rather than eating the same yogurt every day means you’re exposing your gut to different bacterial strains.
Expand your plant variety - Rather than fixating on hitting exactly 30 plants weekly, focus on gradually increasing the variety you’re consuming. Count vegetables, fruits, whole grains, legumes, nuts, seeds, herbs, and spices. Different varieties count separately. If you’re currently at 10-15 different plants weekly, working towards 20 is meaningful progress. The research shows that higher diversity consistently correlates with better metabolic markers, so any increase in variety helps.
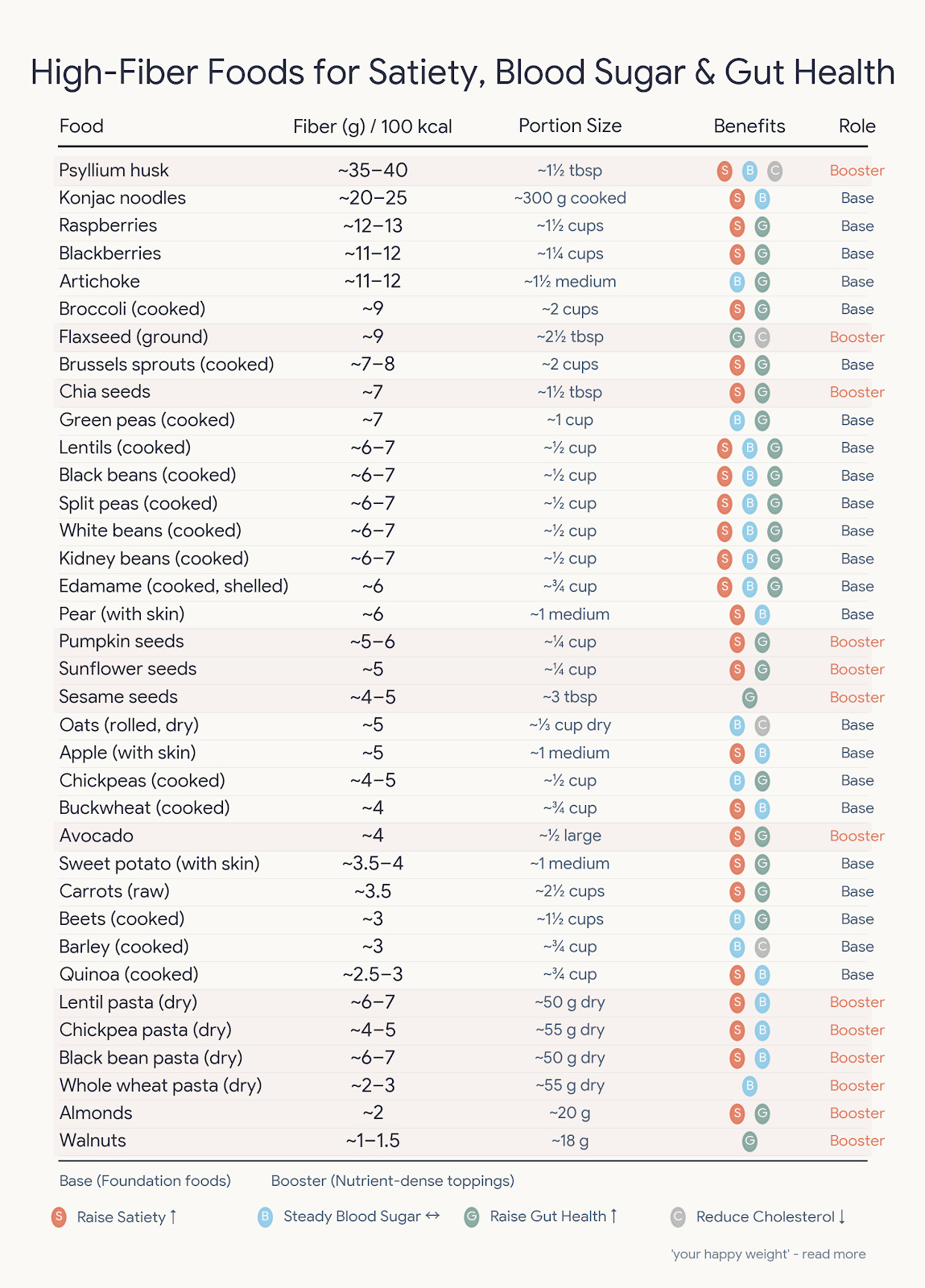
Build up fibre intake gradually - The current recommendations suggest 25g at a minimum. For microbiome effects, research suggests 40-50g may be more beneficial. Consider emphasising soluble and fermentable fibres: oats, beans, lentils, berries, apples, root vegetables, and foods high in resistant starch (cooked-then-cooled potatoes, rice, pasta, green bananas).
Aim to increase your fiber intake slowly over several weeks to avoid digestive discomfort.
Emphasise polyphenol-rich foods - Berries, grapes, green tea, cocoa, olive oil, and deeply coloured vegetables all support beneficial bacteria while inhibiting harmful ones.
Reduce ultra-processed foods where possible- Diets high in ultra-processed foods are consistently associated with less diverse gut bacteria in observational studies. Whether this is because these foods lack fibre and plant diversity, because certain additives directly harm beneficial bacteria, or both, isn’t yet clear from human studies. What we know is that the overall dietary pattern matters - and UPFs typically displace whole foods that support the microbiome.
The payoff isn’t just weight management. Lower inflammation, improved mood, better sleep, and a metabolic system finally working with your efforts rather than against them.
Your gut bacteria are either your biggest obstacle or your strongest ally in reaching a healthy weight. What you feed them matters.



Love this! Will add the list to my bible. (She says while eating a cookie….sigh….)